Human pseudo-embryos created from skin cells present ethical minefield
A team led by Argentine biochemist José Polo has succeeded in generating these models without sperm or eggs but what protection will be they afforded?

A team of scientists led by Argentine biochemist José Polo has succeeded in creating human pseudo-embryos without using eggs or sperm but by reprogramming skin cells to return to an embryonic state. This medical leap, on the shifting frontiers of bioethics, will require humankind to address a pressing question: what legal status will these clumps of human cells, known as blastoids, be afforded?
Polo, of Monash University in Melbourne, prefers to distance his work from the term artificial embryo: “We don’t want to generate false expectations or lead people to think that we’re going to have an army of clones inside a year. The technology has not reached that far yet.”
The biochemist lays out what happens during the first days following fertilization, when the egg and the sperm combine to form a single all-powerful cell, with an instruction manual to turn itself into a human being consisting of 37 billion cells. On the first day, the embryo consists of just two cells. On the second it becomes four. On the third, eight. Sometime around day six it forms into a structure measuring a little more than 0.1 millimeters and comprising of some 200 cells: a blastocyst.
These embryo models, until now, have been unable to generate a complete organism, but who knows if that could be achieved within 10 or 15 yearsSpanish biologist Marta Shahbazi from Cambridge University
“It’s like a football that has a tennis ball inside it,” Polo says. “The football, when it is implanted into the uterus, is what will generate the placenta. And the tennis ball is what will produce the baby.”
Polo’s team has taken skin cells and wound them back to a state similar to that of cells in a natural blastocyst, which are capable of giving rise to a multitude of other specialized cells including blood, liver, muscle and brain cells. When placed in contact, these reprogrammed cells interact and after six days form a human pseudo-embryo, a structure the size of a grain of sand that the team calls an induced blastoid, or iBlastoid. One single experiment can produce thousands.
The researchers managed to grow pseudo-embryos in laboratory conditions to the equivalent of an 11-day-old natural blastocyst. International consensus, based on investigations with surplus human embryos from fertility clinics, establishes the red line at 14 days – the moment when an embryo can no longer divide to potentially produce twins and the current cut-off point for the study of human embryos. As of the 14th day, the concept of the individual comes into play, although the reality is that up to 75% of successful fertilizations are lost in the first two weeks of pregnancy, according to Polo’s data.
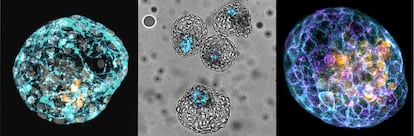
Human pseudo-embryos open the black box of embryonic development – which is difficult to access due to obvious ethical limitations – and allows researchers to investigate the causes of infertility and congenital illnesses. “The iBlastoids can serve as a model for the first two weeks and as such we can study what mutations or what toxins cause the process to fail,” says Polo, whose breakthrough study was published in Nature on March 17.
Another team led by Chinese molecular biologist Jun Wu announced in a parallel paper in Nature that they had succeeded in creating pseudo-embryos from embryonic stem cells derived from natural blastocysts, thanks to the optimization of culture protocols in the laboratory.
This represents the first time that complete models of a human embryo have been created, according to Spanish biologist Marta Shahbazi from Cambridge University. Shahbazi believes that this could be the first step of a “revolution” that will finally unlock the secrets of human development. “These are not embryos, that should be made very clear. They are simply models that can help us to study specific mechanisms that we cannot study in an embryo,” she notes. In 2016, Shahbazi’s team was able to grow human embryos outside of a uterus in laboratory conditions for a longer period of time than anybody had managed to that point: 13 days.
Human pseudo-embryos allows researchers to investigate the causes of infertility and congenital illnesses
The human pseudo-embryos produced by both Polo and Wu’s teams are not functional, that is to say, they degenerate with the passing of the days. Equivalents implanted in the uterus of female rats are also not viable. All the evidence suggests that pseudo-embryos are incapable of producing babies of any kind. Shahbazi believes there is “no ethical problem” in studying these structures beyond the red line of 14 days. “There is no legislation that establishes where the limits are for this type of investigation because it is something very new, but of course limits will be placed upon it. These embryo models, until now, have been unable to generate a complete organism, but who knows if that could be achieved within 10 or 15 years.”
Last year, a team led by Spanish biologist Alfonso Martínez Arias managed to create structures similar to a human embryo of between 18 and 21 days from cells grown in the laboratory, but without the seed for what will be a brain or the tissues that form the placenta. Martínez Arias, then at Cambridge University, is one of the leading figures in the field who in 2018 formed a front calling for debate on the ethical aspects of investigation using models of human embryos.
Martínez Arias believes the two new studies are a “step forward, but not a huge breakthrough” because both models still present functional and structural differences to real embryos, as well as containing foreign cells that should not be there. “The end objective is to substitute the early-stage embryo for these structures that avoid the ethical issue of having to depend on embryos. You can’t just help yourself to human embryos as though you’re buying oranges.”
Now at the Pompeu Fabra University in Barcelona, Martínez Arias believes perfected embryo models that will emerge in the future will have to be regulated under the same ethical guidelines as real embryos. He is, however, in favor of removing the 14-day red line on a case-by-case basis, as long as there is a good scientific justification for doing so. “It has to be done with caution. If everyone is given free rein, I think it will lead to a lot of questionable, headline-seeking science being carried out in place of serious research,” he says.
If we are capable of taking a skin cell and creating an embryo, we are not going to protect skin cells as though they were viable embryosMaría Casado, founder of the Observatory on Bioethics and Law
Research into human embryo models is continuing apace. Wu, of the University of Texas, collaborated in 2019 with the Spaniard Juan Carlos Izpisúa on the creation of artificial rat embryos using a single ear cell. Two other leading laboratories, those of Magdalena Zernicka-Goetzin in Poland and Yang Yu in China, have also presented preliminary results in recent weeks on their work with human embryo models.
María Casado, founder of the Observatory on Bioethics and Law at the University of Barcelona, lent her support to the use of surplus embryos from fertility centers for investigation in 2000. In her view, the key is in the idea of gradualism. “Do we place the same importance on a person, a viable fetus, an embryo in vitro, a germinal cell and a skin cell? The levels of protection need to be gradual; you can’t look for all-or-nothing solutions.” The lawyer believes controversy over the field should be “defused” in favor of research that leads to benefits for human health. “If we are capable of taking a skin cell and creating an embryo, we are not going to protect skin cells as though they were viable embryos,” she states.
Casado is also in favor of revising the current ethical boundary of 14 days: “We cannot attempt to regulate things once and for all, as though it were written down in scripture.”
English version by Rob Train.








































