First RSV vaccine paves the way for ‘new era’ in fight against emerging pathogens
Thanks to years of research into the respiratory syncytial virus, scientists were able to develop Covid-19 vaccines in record time
An experimental respiratory syncytial virus (RSV) vaccine has achieved high efficacy in late-stage human testing for the first time in 60 years, when the search for a vaccine began.
This virus kills around 100,000 newborns and tens of thousands of older people every year around the world. It is a seasonal pathogen that causes pneumonia in adults and bronchiolitis in infants.
The story of the vaccine is a story of scientific discovery. If years had not been spent researching the virus at the molecular level, it would not have been possible for scientists to create the Covid-19 vaccine, which saved millions of lives in just months.
The respiratory syncytial virus vaccine has been developed by the pharmaceutical company GlaxoSmithKline (GSK), and is known by the scientific name RSVPreF3 OA. It has been tested in 25,000 patients over 60 years of age. According to the results of the clinical trial, published in the prestigious medical journal The New England Journal of Medicine, the vaccine has an efficacy rate of 82.6% against respiratory disease and 94.1% against the most severe cases. It also has high efficacy among patients who had weakened immunity due to coexisting conditions. What’s more, the vaccine is equally effective against the two main variants of the pathogen, which can circulate simultaneously.
In addition to the GSK vaccine, which is designed for the elderly, Pfizer has also tested a vaccine for pregnant women. Mothers immunized with the vaccine generate antibodies that cross the placenta and protect the baby from serious infection. It is 81% effective in the first three months of the baby’s life, and 67% in the next three, according to preliminary data published by Pfizer. These two vaccines, the most advanced on the market, come after Europe recently approved a monoclonal antibody that protects children during the first five months of life.
The respiratory syncytial virus was discovered in 1956 and is currently the second most-common cause of death in children under one year of age after malaria. Babies from zero to six months are the most vulnerable, since their immune system still does not know how to fight the pathogen. After two years of age, practically all children have been infected at least once, and successive infections protect them until, in most cases, they are no longer serious.
In 1965, the first vaccine based on an inactivated virus began to be tested. Everything seemed to be working until the winter of 1966, when babies began to become infected. Not only did the vaccine fail to protect them from the virus, it worsened their reaction, increasing their chances of hospitalization. Two babies died. Research in the field was paralyzed for decades.
The reason for the vaccine’s failure would not be uncovered for almost 50 years. In 2013, a team at the US National Institutes of Health, led by Barney Graham, pioneered the use of X-ray crystallography to immobilize the microscopic virus and work out – atom by atom – the complete structure of its F protein, which is essential for the virus to enter human cells and cause an infection. The researchers found that this protein was like a microscopic monster. Before entering a cell it was shaped similarly to an incisor tooth, but later it turned into a sharp fang capable of piercing the cell membrane. This allowed the virus to reach the interior of the cell and hijack its biological machinery to reproduce itself.
The researchers discovered that upon first contact, the human immune system is unable to recognize the viral protein when it is fang-shaped. That is why the first vaccine against the virus was not effective. However, the immune system is capable of identifying the same protein when it resembles an innocent popsicle, and it can efficiently kill the virus before it does any harm. The scientists were the first to succeed in paralyzing the elusive molecule in its pre-fusion configuration, which is the basis of current vaccines and therapeutic antibodies.
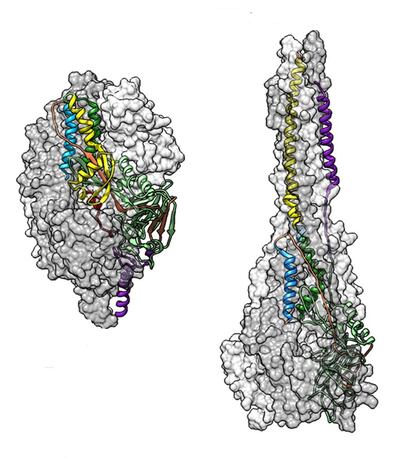
At the beginning of 2020, all those years of work allowed the same team to work out the shape of the coronavirus S protein in just 12 days. Like the incisor-shaped protein, the coronavirus S protein allows the virus to enter human cells and cause an infection. The researchers’ work was essential to the record-fast development of Covid-19 vaccines. “This is kind of the beginning of a new era,” said Graham in an editorial alongside the two new studies.
Federico Martinón-Torres, head of pediatrics at University of Santiago Hospital (CHUS) in Spain, who participated in the clinical trials, believes that “Graham is God.” “Thanks to his technique – the structural development of antigens – these vaccines have finally been achieved,” he told EL PAÍS.
There are still some questions about the RSV vaccine, such as how long immunity will last, whether booster doses will be necessary, and whether it is also effective in people over 80, as the current efficacy data is not conclusive on this point. The GSK trial will continue to investigate these issues.
A vaccine developed by the pharmaceutical company Janssen also reported positive results on Wednesday. In a phase 2 trial involving 5,700 patients, the vaccine was found to reduce the risk of severe disease by 80% among people aged 65 and over. Another two vaccines are also in the third and final phase of clinical trials.
Martinón-Torres believes that the GSK and Pfizer vaccines may be approved by US and European health authorities “in late 2023 or early 2024.” “This may allow us, for the first time, to combat the four viruses that cause severe or even fatal pneumonia with vaccines: influenza, Covid, pneumococcus, and respiratory syncytial virus,” he said.
Azucena Bardají, a specialist in maternal, child and reproductive health at the ISGlobal health institute, also believes that the World Health Organization may recommend the RSV vaccine for pregnant mothers by the end of the year.
Until now, the incidence of RSV among the elderly was unknown, especially among those with weakened immune systems due to other conditions. The advent of the Covid pandemic and the tests required for diagnosis have shed light on its prevalence, showing that RSV can cause as much illness and mortality as the flu, which kills between 300,000 and 650,000 people worldwide every year.
Nearly all (97%) of RSV cases and deaths occur in low- or middle-income nations. While the vaccine may be approved soon in Europe and the US, it is not clear when it will reach these countries.
Sign up for our weekly newsletter to get more English-language news coverage from EL PAÍS USA Edition








































