Potent antifungal antibiotic found in rotten potatoes
Spanish and British researchers discover a bacterium in the tuber that produces a broad-spectrum fungicide
Two things can happen to that forgotten potato at the back of the pantry: either it produces sprouts like in a classroom experiment, or it gets soft, dark and very smelly. In the latter case, it has succumbed to soft rot caused by bacteria. One such bacterium called Dickeya solani (D. solani) was identified in 2005 in northern Europe, and has become a major blight for European potato crops. But a team of scientists recently discovered a silver lining in potatoes with D. solani: a potent broad-spectrum antibiotic that kills fungi competing for the tuber’s nutrients. Laboratory tests have demonstrated that this compound is effective against a wide range of pathogenic fungi, some of them found in humans.
Miguel Ángel Matilla, a molecular microbiologist with the Zaidín Experimental Station in Granada (Spain) collaborated with scientists from the University of Cambridge (UK) to discover this antifungal that synthesizes D. solani. It’s a story reminiscent of the discovery of penicillin in rotten cantaloupes in the mid-1900s that led to the world’s first antibiotic produced on an industrial scale. Like penicillin, this new discovery was the result of applying prior knowledge, scientific curiosity, and the best available technology. “We were studying a different antifungal compound (oocidin A) produced by another beneficial plant bacterium when we discovered the same genes present in the D. solani pathogenic bacterium,” said Matilla.
We were studying a different antifungal compound produced by a beneficial plant bacteriumMiguel Ángel Matilla, molecular microbiologist with the Zaidín Experimental Station
Matilla, who began this research project when he was with the University of Cambridge, and his British colleagues initially grew D. solani mutations that failed to produce the oocidin A fungicide. “We observed that these genetically modified strains were still killing fungi,” said Matilla, “which told us that it was producing a different antifungal molecule.” Using molecular microbiology and genomics, they identified the new compound and named it solanimycin.
The study in mBio, the scientific journal published by the American Society for Microbiology, describes how Matilla and his team discovered how the bacteria produces the solanimycin compound deliberately, almost on demand, in response to cell density. In acidic Ph environments like potatoes, the bacterium activates the group of genes behind solanimycin production. University of Cambridge biochemist Rita Monson calls it an ingenious protection mechanism. “It’s an antifungal that we think works by killing its competitors, an inhibitory process that provides a lot of benefit for the bacteria.” Matilla adds, “If they are able to inhibit or even kill the competing fungi, they will have better access to the nutrients present in the potato plants and tubers.”
It’s an antifungal that we think works by killing its competitors, an inhibitory process that provides a lot of benefit for the bacteriaRita Monson, Department of Biochemistry, University of Cambridge
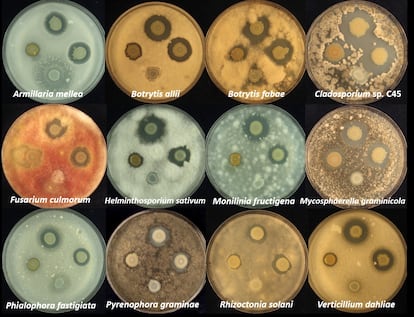
The researchers found that solanimycin works against other substances besides potato fungi. They inoculated the bacterium into about 20 fungal cultures, including several of the most harmful pathogens for various crops. The team also inoculated some yeasts like beer yeast (Saccharomyces cerevisiae), and the Candida albicans fungus found in human respiratory, digestive, and genital tracts that can degenerate into a pathogen in certain conditions. The research team found that the newly discovered compound was effective against most of the plant fungi and yeasts studied.
“It has a broad spectrum of action that is also very specific,” said Matilla. While solanimycin was ferociously effective against fungi, it was not toxic for other microorganisms like bacteria and nematodes. “We don’t yet understand the molecular mechanism of how the compound works, but we think it must interfere with some basic process in fungi,” he said.
Before solanimycin can be used as a drug, several years of work lie ahead to study its molecular structure and better understand how it works. After the compound has been identified, it will either have to be purified in large quantities or a synthetic version must be developed. Following that, it must be used in cellular models to determine its toxicity and later in plant or animal models before it can be tested in clinical trials.
Even if solanimycin is never turned into a drug, researchers believe that its discovery has already achieved one goal. Up to 80% of antibiotics (including antifungals) are derived from soil microorganisms. The vast majority of these are actinobacteria in drugs that have been administered to humans so frequently that a new problem has arisen – the development of resistance. Matilla says that the discovery of solanimycin produced by potato-rotting bacteria shows that “we must also look to the plant microbiome as a source of new antibiotics.”








































