Three paralyzed patients walk again thanks to spinal cord implants
A Swiss team used artificial intelligence software to send signals imitating the brain’s instructions to the lower limbs, letting people take steps independently

Three people who had been left paralyzed in motorcycle accidents have been able to walk again thanks to a system developed by Swiss researchers involving implants that are placed directly on the spinal cord.
All three individuals had lost their trunk and leg motor functions due to complete spinal cord injuries. “A day after I started to train I saw that my legs were moving again; it was a very intense emotion,” said Michel Rocatti, one of the three patients, at a news conference.
The neuroscientist Grégoire Courtine of the Swiss Federal Institute of Technology (EPFL) in Lausanne, and the neurosurgeon Jocelyne Bloch of Lausanne University Hospital (CHUV) directed the scientific team that managed the feat.
In a four-hour operation, doctors attached electrodes that send out synchronized electrical signals imitating the signals that normally run through the spinal cord to deliver the brain’s instructions to the lower limbs. These implants are connected to a computer with an artificial intelligence software that reproduces the necessary impulses to allow the patient to stand, walk and even perform more complex activities such as riding a special bicycle and swimming. The achievement was described in a paper published on Monday in Nature Medicine.
This new milestone adds to progress made by two teams in the United States that used continuous electrical stimulation to make paralyzed patients walk again. The main idea that emerges from this work is that some forms of spinal cord injury are no longer irreversible.
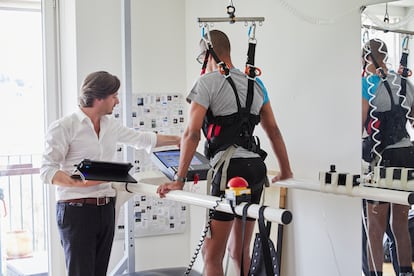
Courtine’s team has been trying to make paralyzed people walk again for years. In 2014 the researchers tested their system on mice with severed spinal cords, and two years later it did the same with monkeys. In the fall of 2018, the Swiss team showed its progress with David Mzee, a young man who was in a wheelchair after suffering a partial spinal cord injury at age 20. Thanks to electrical stimulation of the spinal neurons, Mzee was able to stand and take a few steps with a walker.
In the latest trial, the electrodes and their long cables were crafted specifically for the test, taking each patient’s injuries into account. “Until now, all these kinds of implants were reutilizing electrodes originally designed to treat pain,” explained Courtine. “Designing for the first time a specific technology for this new use allows us to better synchronize the stimulation with the movement itself, imitating the real signals that the brain sends out while walking, for instance.”
On this occasion, scientists managed to stimulate not just the spinal neurons in charge of leg motion, but also those regulating the abdomen and lower back muscles. Patients were able to stand up soon after their surgery, and took their first steps in a harness. It required a lot of training to refine their movements, but after four or five months Rocatti was able to go out for a walk in the street and have a drink standing at a bar. He now uses a walker that lets him control the intensity and pace of the electrical impulses. “When I use the device I feel better, stronger, and the pain associated with the wheelchair goes away,” he said.
The Swiss team has treated nine people with the experimental system. Courtine said that his team is hoping to start clinical trials with more patients in 2023, partly through Onward Medical, a company he has founded with Bloch to commercialize the technology in the future. The trials will still take a few years of work. “We are going as fast as we can,” said the neuroscientist.
When I use the device I feel better, stronger, and the pain associated with the wheelchair goes awayMichel Rocatti, patient
“These new results are spectacular,” said Filipe Barroso, a researcher at the neuro-rehabilitation group at the Cajal Institute, part of Spain’s National Research Council (CSIC). According to him, the most notable achievement is the fact that these three patients had complete spinal cord injury, instead of a partial one as with the 2018 patients, who still preserved some residual functions. “And the results became apparent in just one day, which can be explained by an optimal placement of the electrodes,” he added.
Barroso is working on less invasive stimulation systems that can be placed on the skin or inside the muscles. In the latter case, his team showed last summer that electrodes can achieve “a spectacular” reduction of shaking in patients with nervous system conditions.
Diego Serrano, a researcher at Castilla-La Mancha University in Spain, visited Courtine and Bloch’s lab in 2018. “It is obvious that they are refining their technique in order to achieve a movement that is as natural as possible,” he said. “The electrodes are now implanted with pinpoint accuracy, and the fact of placing them one or two millimeters higher or lower makes a huge difference.”
But Serrano also said it will be complicated to use this technology on a larger number of patients. “It’s difficult because each spinal cord injury is very specific, practically unique, which means you need to develop a specific treatment for each person.”
Tu suscripción se está usando en otro dispositivo
¿Quieres añadir otro usuario a tu suscripción?
Si continúas leyendo en este dispositivo, no se podrá leer en el otro.
FlechaTu suscripción se está usando en otro dispositivo y solo puedes acceder a EL PAÍS desde un dispositivo a la vez.
Si quieres compartir tu cuenta, cambia tu suscripción a la modalidad Premium, así podrás añadir otro usuario. Cada uno accederá con su propia cuenta de email, lo que os permitirá personalizar vuestra experiencia en EL PAÍS.
¿Tienes una suscripción de empresa? Accede aquí para contratar más cuentas.
En el caso de no saber quién está usando tu cuenta, te recomendamos cambiar tu contraseña aquí.
Si decides continuar compartiendo tu cuenta, este mensaje se mostrará en tu dispositivo y en el de la otra persona que está usando tu cuenta de forma indefinida, afectando a tu experiencia de lectura. Puedes consultar aquí los términos y condiciones de la suscripción digital.









































