Spanish scientists help blind woman to see forms with brain implant
The groundbreaking project enabled a biology teacher to recognize shapes and letters, and even play a simplified version of Pac-Man
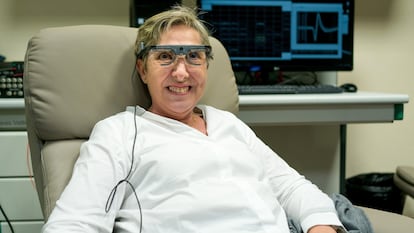
Sixteen years ago, Bernardeta Gómez, 57, was teaching biology at a high school in Valencia when toxic optic neuropathy destroyed the bundle of nerves that connect her eyes to her brain, leaving her blind. Now, an implant inside her brain has enabled her to make out patterns and recognize various letters of the alphabet. She has even been able to play a simplified version of the Pac-Man computer game.
Designed by scientists at the Miguel Hernández University of Elche (UMH) in the Spanish province of Alicante, the system consists of a 96-electrode implant that is placed in the rear brain region responsible for vision. This is the first time this technology has been used on a blind person. The former teacher’s blindness returned once she was unhooked from the system, but now new volunteers are being recruited to take the experiment further.
“I got the implant on Monday, October 22, 2018,” Gómez recalls. “On Tuesday, we were already embarking on the experiment. During the first three months, which was the [initial] time frame for the research, we didn’t make much progress, so I insisted that it be extended. It was then that I began to perceive changes in intensity. I could see something like very luminous sequins and, as the parameters varied, I saw them in different degrees of intensity and size.”
What Gómez calls “sequins” are actually phosphenes, a visual phenomenon in the form of flashes or luminous white-yellow dots. Anyone who rubs their eyelids with a certain degree of pressure will see a shower of them. In blind people, they are common and often unprompted. In Gómez’s case, she used to see them when there was a loud sound or she was startled, but also sporadically, without triggers.
Eduardo Fernández, the director of the Biomedical Neuroengineering Group of the Institute of Bioengineering at UMH, explains that phosphenes appear at a specific point in the visual sphere. “The retina has a kind of map in the cerebral cortex that connects with the visual field,” he says. “This retinotopic map had been studied in sighted people: you stimulate a certain part and you see something specific and not other things.” Fernández’s team was surprised that the same thing happened to a blind person. “The map is still there,” he says.
I started to see narrow bars, wide bars, squares and then I learned to distinguish patternsBernardeta Gómez, volunteer in experiment
The problem they encountered with Gómez is that sometimes these phosphenes appeared when stimulating the right occipital cortex [behind the ear, at the top of the brain], where the seat of visual processing lies, and at other times they did so without a catalyst. As Gómez observes wryly, “They [the scientists] were as blind as I was.”
The research, that has been carried out in collaboration with scientists from the Netherlands’ Institute of Neuroscience and the University of Utah in the United States, is pioneering in many ways. The researchers inserted a plate with 96 electrodes inside the brain. Each plate is 1.5 millimeters long and 80 microns in diameter. “They are the size of the neurons we want to communicate with,” Fernández explains. Neuroscientists have been testing these plates for several years on people who are paralyzed or unable to communicate. “It is the first time that one has been implanted in the visual region of the brain and in a blind person,” he adds. The electrodes not only send electrical signals; in a groundbreaking development, they also send the neuronal response to an external system. All the details of the study appear in the latest edition of the scientific journal, Journal of Clinical Investigation.
The system was completed with an artificial retina – an image processor similar to that of a camera, mounted on a conventional pair of glasses. Its mission was to substitute the optical stimulus for an electrical one to activate the brain. Unlike other approaches, such as optogenetics that try to get the eye to recover part of its vision, “here, we bypassed the eye,” says Fernández. Under normal circumstances, Gómez’s eyes wouldn’t even allow her to perceive light.
To begin with, the researchers activated the electrodes one by one, thereby triggering the appearance of a single phosphene. Gómez remembers telling them, “I’m seeing a dot.” As her brain became educated, the researchers increased the complexity of the stimulation, ramping up the number of electrodes they activated at a time. “They made it more complex and I started to see narrow bars, wide bars, squares... and then I learned to distinguish patterns. I got to perceive a human face and the face of a dog.” She was even able to play a simplified version of Pac-Man in which she had to avoid getting caught. “It was – how should I put it – a real experience,” she says. “I’m at a loss for words.”
In the detection of patterns and shapes, the patient went from a success rate of 81.4% to 100%. In the last month of the six-month experiment, the team went further and investigated her perception of letters using the activation of 16 electrodes simultaneously. Gómez was able to distinguish some, such as L, C, V and O, with a 70% success rate. But they could not induce the perception of the entire alphabet and are not sure why.

Almost everything in the research is so new that there are no precedents to compare it with. For example, they had to keep adjusting the electrical signal until they identified the threshold needed to achieve a response from Gómez’s brain. Attempts to stimulate the brain to help blind people regain at least some vision date back to the 1970s. But it was always stimulation from the outside. In this case, they open the head and go directly into the brain. Unlike electrode systems placed on the scalp, which operate in the range of a milliampere –an ampere being a measure for small electric currents – these direct implants significantly lower the amperage. The proximity allows for higher resolution with less current, but it was necessary to finetune the system to avoid excessive stimulation. Gómez’s average threshold was put at 66.8 microamperes.
According to Jaume Català, ophthalmologist at the Catalan hospitals, Sant Joan de Déu and Bellvitge, the results of this research are “a milestone in artificial vision with cortical stimulation.” But he promptly tempers his enthusiasm, explaining: “This is an individual case and a pilot study.” The approach, he says, is capable of proposing potential solutions for patients who once had vision and have lost it completely, either due to retinal or optic nerve alterations. But, he adds, the visual cortex must be functional. “We are still far from achieving functional vision because of the implants’ durability and predictability, and the need to study and better understand the complex and multiple pathways of the visual areas of the cortical region,” he says.
The research also serves to underscore how much remains to be done in the field. For people like Gómez to be able to take advantage of the results of research of this type, several problems still need to be solved. One is the number of electrodes. The 96 used here, which occupy a square measuring 4x4 millimeters, would have to be greatly increased. Fernández’s team published an experiment in the journal Science in which they used a plate with 1,024 electrodes to study brain response, “but it was on monkeys that could see,” he says.
Another problem is that Gómez had to be connected to a central unit while participating in the tests. Once she was done, she would unhook and stop seeing the phosphenes. Transmitting and receiving wirelessly would require the kind of electrical current that could create other problems. “We need more data,” says Fernández. In a bid to get more, they have extended the trials to other volunteers. “We have received requests from all over the world from people hoping to see again, but that is not going to happen; this is research. We are looking for people like Berna, who know they are not going to see again. Otherwise, we could do them a great deal of harm.”
Bernardeta Gómez insists that she does not feel particularly nostalgic about having been able to see certain things again over the space of a few months. She explains: “I knew where I was going with this. It was very clear to me that I wasn’t going to recover my sight, but I feel a great personal satisfaction and that’s my compensation.”
Tu suscripción se está usando en otro dispositivo
¿Quieres añadir otro usuario a tu suscripción?
Si continúas leyendo en este dispositivo, no se podrá leer en el otro.
FlechaTu suscripción se está usando en otro dispositivo y solo puedes acceder a EL PAÍS desde un dispositivo a la vez.
Si quieres compartir tu cuenta, cambia tu suscripción a la modalidad Premium, así podrás añadir otro usuario. Cada uno accederá con su propia cuenta de email, lo que os permitirá personalizar vuestra experiencia en EL PAÍS.
¿Tienes una suscripción de empresa? Accede aquí para contratar más cuentas.
En el caso de no saber quién está usando tu cuenta, te recomendamos cambiar tu contraseña aquí.
Si decides continuar compartiendo tu cuenta, este mensaje se mostrará en tu dispositivo y en el de la otra persona que está usando tu cuenta de forma indefinida, afectando a tu experiencia de lectura. Puedes consultar aquí los términos y condiciones de la suscripción digital.








































