Spain approves Covid-19 vaccines for children, with drive set to begin on December 15
The child dose of the Pfizer-BioNTech medication will be administered to the five to 11 age group, which has the highest incidence rate of the coronavirus in the country by demographic
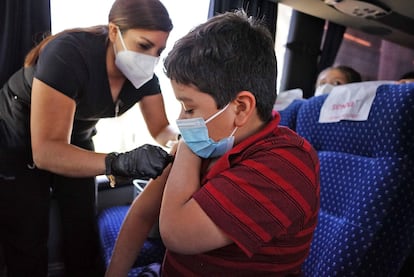
Spain will begin to vaccinate children aged between five and 11 years old against Covid-19 from December 15. On Tuesday, the country’s Public Health Commission approved the inoculations among the under-12s after the Pfizer-BioNTech vaccine for this age group was validated by the European Medicines Agency (EMA), and was backed by the group of experts that is advising the Spanish health authorities.
The campaign will begin as soon as the first vials arrive in Spain, something that is due to happen on December 13. From there, the vaccines will be distributed among the country’s regions, which will be able to begin the process from December 15 onward. In Spain, each of the 17 territories is in charge of its own healthcare systems, vaccination programs and coronavirus restrictions.
The Health Ministry is expecting to receive 3.2 million doses during December and January, which will be sufficient to administer a first shot to practically all children in Spain aged between five and 11, who number 3.3 million. The second dose will be administered eight weeks later.
The under-12s are the only age group not to be included in Spain’s ongoing vaccination campaign, and is also the group with the highest incidence of cases. According to the latest Health Ministry report, there were 412 cases per 100,000 inhabitants in this age group over the previous 14 days, nearly double the average for Spain (248) and more than triple that for adolescents (133) or the over-80s (125). After children, the groups with the highest incidence rate are the 30- to 50-year-olds, which coincides with the ages of most parents of these youngsters.
These figures are among the main reasons why public health chiefs at the ministry and in the regions have opted to approve vaccines for the five- to 12-year-old group (no Covid-19 vaccines have been approved for the under-fives as of yet). Just as the fifth wave ahead of summer spread rapidly among teenagers, who were not vaccinated at the time, in this, the ongoing sixth wave of the pandemic in Spain, the virus has spread fast among children.
There is no urgency, but we have too many cases and children are contributing to transmission in the countryQuique Bassat, epidemiologist and member of the Spanish Pediatric Society
While the Covid-19 vaccines do not stop transmission altogether, they do reduce it, meaning that once children have been fully inoculated it is likely that the spread of the virus will slow (not taking into account what could happen with the recently discovered omicron strain, which is apparently more transmissible).
Clinical trials have shown that with the other strains of the coronavirus that are circulating, the Pfizer vaccine for children – which has a third of the dose administered to adults – reduces the probability that a child will contract the disease by 91%. It also minimizes the already low chances of hospitalization, need for intensive care or death to practically zero.
In a context of low transmission, the specialists consulted by EL PAÍS were not in favor of vaccinating children, given the low risk of serious cases of Covid-19 among this age group. But the higher the curve, the more vaccination for children makes sense. That was the conclusion, for example, of the group of experts advising the United States Food and Drug Administration (FDA), which found that the benefits of vaccinating children is greater than the risks in any scenario, but in particular in one of high contagion rates.
Epidemiologists consulted by this newspaper back the new plan. “For their protection, but in particular to slow transmission,” stated Fernando Rodríguez Artalejo, a professor in Public Health at Madrid’s Autonomous University. The experts see this as one of the weapons that will help with the fight to corner the virus.
Quique Bassat, an epidemiologist and member of the Spanish Pediatric Society, said that without being urgent, vaccines for children are starting to be necessary. “The data backs this,” he said. “The pediatric group has the highest incidence by far, there are outbreaks in schools, something that had not happened until now. There is no urgency, but we have too many cases and children are contributing to transmission in the country.”
The debate about child vaccination, however, is different to that involving adults, whose chances of dying from Covid-19 are much greater. Among children, serious side effects from the vaccines – while very rare – can be more so than those caused by the illness itself. While there were no serious side effects among the under-12s during clinical trials, among adolescents there were very sporadic cases of myocarditis, which in most cases were resolved with conservative intervention and without the need for intensive care treatment.
Experts in the US concluded: “While benefits of vaccination were highly dependent on COVID-19 incidence, the overall analysis predicted that the numbers of clinically significant COVID-19-related outcomes prevented would clearly outweigh the numbers of vaccine-associated excess myocarditis cases over a range of assumptions for COVID-19 incidence.”
Vaccination of a healthy child is good, we know that the vaccine works and that it is safe and effectiveFederico Martinón, vaccine consultant for the World Health Organization
The majority of complications from Covid-19 in children occur in those with preexisting conditions. For this reason, Federico Martinón, a vaccine consultant for the World Health Organization (WHO), believes that the most vulnerable children should be involved in the vaccination campaign. This includes youngsters with cancer, suppressed immune systems, obesity, Down syndrome, or those with serious neurological conditions. With respect to the rest, the decision is “complex, and depends on scientific, ethical, political, epidemiological and economic factors.”
“People confuse it being good or bad with whether it is a priority or not,” Martinón added. “Vaccination of a healthy child is good, we know that the vaccine works and that it is safe and effective. And as well as the clinical trials, it has already been administered to more than five million children of that age, with no safety problems. That said, it is not a priority in comparison with the indication of vaccination with other risk groups. Right now I would prioritize the vaccination of all children with risk factors. In the second instance, of children living with adults who are at high risk, in order to create an additional security cordon around those who are most vulnerable. And lastly, would come the vaccination of the remaining children, who are not a priority – which is not the same as [the vaccination process] not being good or not having any data,” he said.
The United States is one of the countries that has already started the process of vaccinating children, with more than 2.6 million doses administered so far to 10% of the population aged five to 11. A month later, no notable side effects have been reported. “We are seeing a good safety profile,” said Bassat. “The cases of myocarditis, which had a lot to do with the dosage, will probably be greatly reduced with the pediatric dose.”
China has just started the biggest campaign in the world, with plans to vaccinate 160 million children before the end of the year. In Europe, after the EMA’s approval, countries such as Italy and Germany will begin vaccinating children once the doses arrive. In the case of France, youngsters with chronic illnesses or other vulnerabilities will be eligible for their doses from December 15 onward. The remainder, some six million children, will be able to do so once the health authorities grant their final approval, likely “around December 20,” according to French health chief Olivier Véran. In the United Kingdom, the process is being studied by a vaccine committee, which may approve the vaccinations for children this week.








































