Elon Musk claims Neuralink has implanted first brain chip in a human
The controversial entrepreneur announced his company’s product will be called Telepathy, and will allow patients to control a cell phone and a computer with their mind


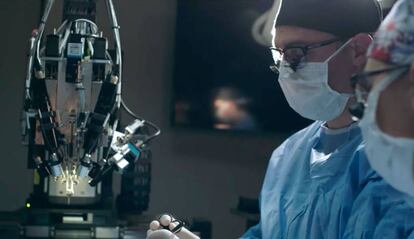
It took a little longer than advertised, but Elon Musk confirmed Monday that a human has received the first brain implant of a chip designed by his company Neuralink, and was “recovering well,” the entrepreneur said as he broke the news on X, the social network he also owns. “Initial results show promising neuron spike detection,” he added. The surgical procedure was performed by Neuralink, which has been under the spotlight of the authorities after the deaths of several monkeys in the experimental phase of the process. Neither the age nor the identity of the patient has been revealed.
Musk has made the technological and scientific breakthrough appear like a marketing milestone. “The first Neuralink product is called Telepathy,” the entrepreneur posted on X. This will serve to allow the patient who has received the implant to control a cell phone and a computer with their thoughts. “Initial users will be those who have lost the use of their limbs. Imagine if Stephen Hawking could communicate faster than a speed typist or auctioneer. That is the goal,” Musk added. This is not the first time brain implant surgery has been performed, as neurotechnology has been advancing in this field for years.
The company began recruiting patients for the six-year experiments in September. These would form part of PRIME, a program that encapsulates the company’s goals: the precise implantation of a chip (Precise Robotically Implanted Brain-Computer Interface) in the brain. In its first major presentation to the public, the company claimed that the implant, which would be “cosmetically invisible,” would allow control of a cursor or keyboard using “thoughts alone.” The search for patients began at the same time that the Physicians Committee for Responsible Medicine, a Washington-based nonprofit organization, together with journalistic investigations, warned of the death of a dozen monkeys during Neuralink’s experiments and lodged a complaint with the U.S. Securities and Exchange Commission.
Neuralink’s objective was to focus on quadriplegics with spinal injuries, or people suffering from amyotrophic lateral sclerosis (ALS) for a period of at least a year. Those selected had to be aged over 22 and have a nurse or on-call nurse on a continuous basis. They would receive financial support to travel to the clinical trial sites during the 18-month first phase. The big reward, however, would be a radical change in their lives brought about by a chip implanted in the part of the brain that controls the intention to move. The semiconductor records neural activity through 1,024 electrodes distributed over 64 wires, each thinner than a human hair.
Neuralink green light
Neuralink got the green light from the Food and Drug Administration (FDA) to carry out the procedure in humans in May 2023, a prerequisite for the historic step Musk announced on Monday. Health authorities had rejected the company’s first request, made in 2022, due to concerns about the safety of the lithium battery included in the semiconductor, which is the size of a quarter. The government agency’s experts also wanted to know whether the wires coming out of the brain could disrupt or damage other areas inside the skull.
The team at Neuralink, which was founded in 2016, corrected the FDA’s observations in record time and against the opinions of experts, who were skeptical about the procedure. “Neuralink doesn’t appear to have the mindset and experience that’s needed to get this to market anytime soon,” Kip Ludwig, former program director for neural engineering at the U.S. National Institutes of Health, told Reuters in March last year. Musk’s company was in a race to beat its competitor, Paradromics, an Austin, Texas-based company that had developed a similar technology that allows paralyzed patients to regain some abilities.
In 2021, Neuralink released a video of a monkey playing Pong, the Atari console video game that mimics the dynamics of table tennis. For the first time, the company was able to clearly communicate how its implant could revolutionize the future. The novelty was not that the primate, named Pager, interacted with the game, but that it did so by manipulating the controller with its brain after it had been implanted with two chips six months earlier. Musk announced in late 2022 that Neuralink would begin human trials in mid-2023. Today they are a reality, according to the entrepreneur.
Sign up for our weekly newsletter to get more English-language news coverage from EL PAÍS USA Edition







































