The immortal jellyfish reveals its secrets for eternal life
Researchers at the University of Oviedo have sequenced the animal’s genome. The results could shed light on aging and cellular deterioration in other species, including humans
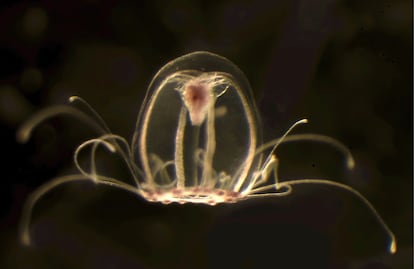
Some animals, like turtles and bowhead whales, are known for their long lifespans. Others, including elephants and naked mole rats, are known for being immune to cancer. Planarians regenerate even after their heads are cut off, and the microscopic tardigrades can survive any catastrophe imaginable. But unique in their abilities are certain species of jellyfish, who, once they reach adulthood, can turn back and become young again. Now, researchers at the University of Oviedo in northern Spain have sequenced the animal’s genome, revealing the keys to their biological immortality. The results could shed light on aging and cellular deterioration in other species, including humans.
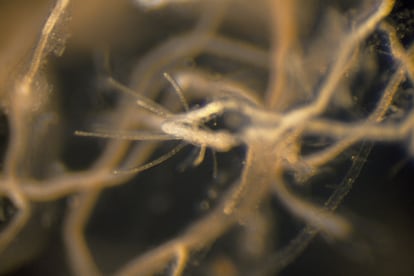
The Turritopsis dohrnii is a small jellyfish that can be found from the Pacific Ocean to the Mediterranean to the Caribbean. It belongs to the anemone and coral family. Many species in this group have cellular regeneration abilities that humans envy, but the T. dohrnii surpasses them all. Under normal conditions, its life cycle is divided into four parts. The union of the male and female gametes produces a larva or planula. It then attaches to the seabed as a polyp, similar to an anemone. The polyps then release ephyra, or young jellyfish in the phase prior to sexual maturity. They become jellyfish, reproduce sexually and start over. But if they are stressed by an environmental threat, the jellyfish revert: after reproducing, they return to the previous phases, becoming polyps on the ocean floor again.
As far as scientists know, jellyfish can repeat the process indefinitely. Thus they are said to be biologically immortal. They can be eaten by a predator or fall into the hands of a swimmer, but they do not die of old age.
Scientist Carlos López Otín, at the University Institute of Oncology of the University of Oviedo, decided to study the T. dohrnii because of its immortality. It is not the first time that his laboratory has scrutinized an animal’s genes: in 2018 they sequenced the genome of George, the last of the Galapagos giant tortoises, who died a decade ago after living about 100 years. They also participated in deciphering the genetic code of the bowhead whale and the tardigrades. The new study sequences the genome of the T. dohrnii jellyfish for the first time and compares it to that of a close relative, the deadly T. rubra. The results, published in the scientific journal Proceedings of the National Academy of Sciences (PNAS), show why the former has earned the title of immortal jellyfish.
Marine biologist María Pascual is a co-author of the study. With the help of the Gijón aquarium, the scientists set up fish tanks in the laboratory to house the jellyfish, which they fed crustaceans. They planned to sequence the genome at different times in the life cycle as the animal grew. “If there was a genetic change during the reversal, it would be important,” she says. They didn’t just see one change. They saw several, all related to DNA replication and repair. Such changes appeared in genes involved in oxidative stress. The researchers also made new findings related to various aspects of cellular aging, such as telomere length. “What makes this animal special is the synergy of all these changes, which make this jellyfish rejuvenate,” says Pascual.
On its way back, the adult jellyfish shrinks and changes the structure of its tissues in parallel. The scientists observed that at the genetic level, some genes stopped expressing themselves –that is, generating proteins and their functions– while others were activated. Such is the case of the GLI3 gene, which is involved in the differentiation of pluripotent stem cells into other types of cells. T. dohrnii has twice as many copies of this gene as T. rubra, and all are active at the time of reversion. This reversal of the cell differentiation process is known as transdifferentiation. It is an extremely rare phenomenon in nature. “The cell returns to the zero point,” says Pascual.
Dido Carrero is a molecular biologist at the Asturias Marine Observatory and co-author of the research. “This jellyfish is the only animal capable of reverting from an already differentiated state in adulthood,” she says. Other jellyfish are capable of reverting their life cycles, but only before they reach sexual maturity. For Carrero, T. dohrnii’s power lies “in the activation of genes related to the state of pluripotency of cells.” From the start, the animal has a higher number of copies of genes involved in cell replication and DNA repair than T. rubra. This duplication means that the first jellyfish has “more efficient cellular mechanisms,” write the authors of the study, than the second.
The senior author of the research is López Otín, who has spent 35 years investigating the cellular mechanisms of cancer. “What I have learned in this time is that cells can become selfish, wandering and immortal. Understanding cancer requires studying other mechanisms and processes of immortality. Hence our interest in this jellyfish,” he explains. The professor has also learned that “immortality is undesirable, aging is inexorable, but longevity is extraordinarily plastic and T. dohrnii carries this plasticity to the limit.”








































