Medical breakthrough: ALS patient in completely locked-in state ‘speaks’ thanks to brain implant
This is the first time the technology has been able to help a person who cannot even move their eyes. Following the procedure, the 34-year-old was able to spell out his name, ask for a beer and suggest a movie to his son
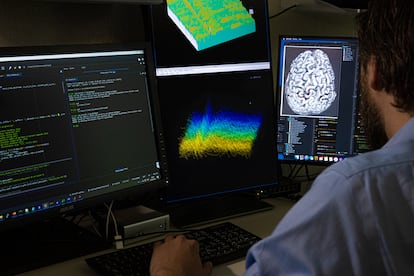
He couldn’t even move his eyes – at that point, his body had been completely immobilized by amyotrophic lateral sclerosis (ALS) – but he was able to say his name, his wife’s and that of his four-year-old son. He didn’t speak with his voice, but rather with his thoughts thanks to a brain implant. Spelling out his name, letter by letter, took some time, but it marked a medical breakthrough: for the first time an ALS patient in a completely locked-in state (CLIS) was able to communicate.
“That moment was truly magnificent,” explains Arnau Espinosa, one of the neuroscientists behind the milestone in Germany. It’s a case that has opened the door to allow other patients with serious conditions to communicate, and taken the development of brain-machine interfaces one step further.
Just 100 days before the feat, doctors at Munich Hospital had inserted two brain implants, with 86 electrodes each, into the patient in order to identify his neural activity. These brain waves are what allowed the 34-year-old, whose name has not been revealed to protect his privacy, to communicate with his family. The patient retains his mental abilities and is able to listen: that’s how he receives the stimuli. After a period of calibration and learning, he was able to use his brain to mentally select letters on a virtual keyboard.

In this way, he could communicate – letter by letter – if he was experiencing pain, discomfort or other medical issues. What’s more, he could communicate with his family. For example, once the implant had been in place for 253 days, he was able to ask his son if he wanted to watch the Disney movie Robin Hood. He was also able to ask for a beer in his feeding tube, to choose potato curry to eat and to request that an album of heavy metal band Tool be played at top volume.
“They are very difficult projects,” says Espinosa, a scientist at the Wyss Center, a neuroscience research facility based in Geneva. “But one of the nicest moments for me was when we let the patient communicate freely, and he spelled out his name correctly: that is absolute evidence that the patient is aware, that he can correctly understand the world around him: I want to say my name and I am saying it.”
The scientific team suggested the brain implant when he was still able to control the movement of his eyes and could communicate via blinking, the same method used by the now-deceased British physicist Stephen Hawking, who also suffered from ALS. The operation was carried out in March 2019. An opening was made in his cranium to create an exterior connector, which communicated with equipment installed in his home.
It’s not the first time this system has been used, but it is the first time it has been used with a patient at this stage of ALS, when they are in a completely locked-in state. Patients with CLIS are cognitively intact, but their brain signals do not reach the nervous system, meaning they cannot control any bodily function and even need a respirator to breathe.
The patient learned he was going to be a father after he was diagnosed with ALS, but that did not deter him from parenthood, says Espinosa. “He knew that the experience that he would have with him was going to be an atypical situation.”
The researchers published the results of the study in the journal Nature Communications. The paper reflects more than two years of work, including the period affected by the coronavirus pandemic, when the scientists had to work with the patient remotely.
“This study responds to the age-old dilemma of if people in a completely locked-in state, who have lost voluntary muscle control, including the movement of the eyes or the mouth, also lose the brain’s ability to generate communication demands,” explains Jonas Zimmermann, a neuroscientist at the Wyss Center.
According to the foundation, more than 300,000 people are expected to be living with ALS in 2040, and many of them will lose the ability to talk. With further development, it is hoped this kind of spelling interface will be able to be used with other patients in the advanced stages of degenerative diseases.
But it is a complex process and the technology still has a long way to go. After a successful period of free communication, the patient’s abilities began to wane, says Espinosa. The system has lost sensitivity, meaning he can longer spell out words, and is limited to replying yes or no to close-ended questions. “The results are declining a little, it’s likely a technical problem,” he explains. “The brain is trying to protect itself from a foreign body and is creating a protective barrier against the electrode. What’s more, it is adapting and the signal from the neural networks is much weaker.”
It will also be some time before these advances become commercially available, says Espinosa. “These implants are far from being a technology that can be applied generally: they are prototypes, concept tests, much more work must be done in order to have a commercial device on the market,” he explains. It’s a field however, that is already attracting billion-dollar investments from companies such as Neuralink, which is owned by Tesla founder Elon Musk.
While part of the technology can be applied to any patient, where the implant needs to be placed varies according to the person. What’s more, the system not only needs to be calibrated to each brain, but also before every session. Espinosa explains: “You can’t plug something in and hope that tomorrow it will be the same way. We are trying to automate it as much as possible. But there is a lot of work to do to ensure the systems and the algorithms adapt to the brain signals from session to session.”








































